Potential Cancer Breakthrough: Groundbreaking New Report Shows 84% Clinical Benefit Ratio for Ivermectin+Mebendazole in Cancer Care
Clinical Research · Oncology
Doctors from The Wellness Company have completed a first-of-its-kind human observational report examining the use of Ivermectin and Mebendazole in the treatment of cancer. The manuscript is currently under peer review.
"Off-label use of Ivermectin and Mebendazole showed a remarkable 84% Clinical Benefit Ratio in the treatment of cancer. These results indicate that the inexpensive and safe off-label applications of these medications could be an important complement in the treatment of cancer."
— Dr. Peter McCullough, Chief Scientific Officer, The Wellness CompanyStudy Overview
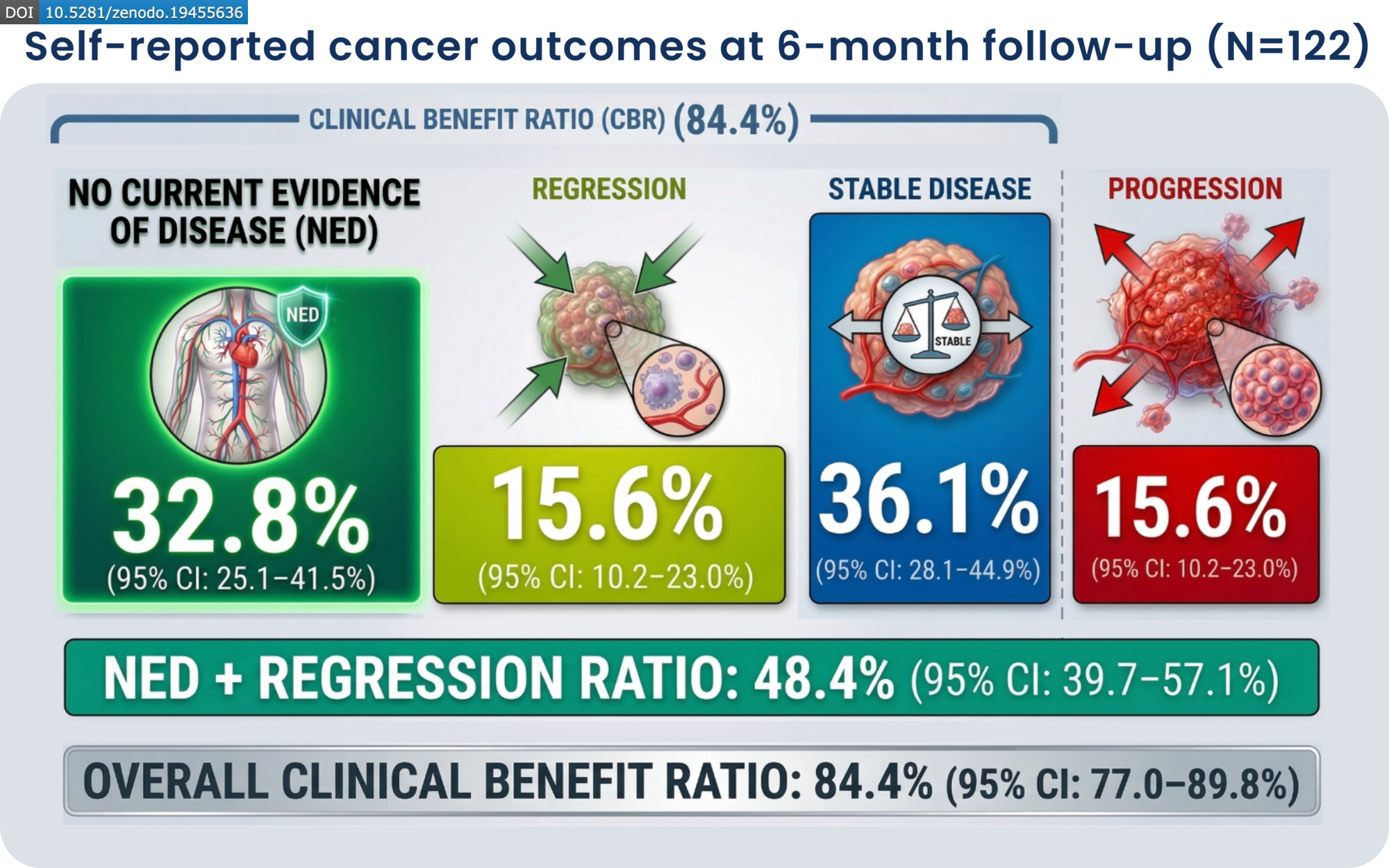
The report analyzed 197 cancer patients prescribed ivermectin and mebendazole off-label. Participants received compounded oral capsules containing 25 mg of ivermectin and 250 mg of mebendazole. After six months, participants reported outcomes that researchers describe as clinically significant.
Only 15.6% of participants reported disease progression. The cohort represented a heterogeneous population of cancer patients, making the breadth of these preliminary findings particularly noteworthy to the research team.
What the Researchers Are Saying
"In this prospective real-world cohort, the combination of ivermectin and mebendazole was associated with high rates of self-reported clinical benefit, with nearly half of participants reporting tumor regression or no current evidence of disease."
— Dr. Peter McCulloughDr. Kelly Victory, cancer survivor and co-designer of the analysis, underscored the significance of real-world data: "Prior to this data, ivermectin and mebendazole have demonstrated highly promising anti-cancer activity in preclinical models. But despite compelling preclinical data and documented safe use in cancer patients, robust clinical evidence evaluating the ivermectin–mebendazole combination in oncology remained limited — which is why this analysis was conducted."
A Call for Urgent Investigation
Dr. Harvey Risch, Professor Emeritus of Epidemiology at Yale School of Public Health and Chair of the President's Cancer Panel at the National Cancer Institute, weighed in on the findings.
"This report reveals an exciting new potential that should expand the consideration of ivermectin and mebendazole for inclusion in the treatment of multiple cancer types. We urgently need a full-fledged scientific investigation into this class of medications and their impact on cancer treatment."
— Dr. Harvey Risch, MD, PhD — Yale School of Public HealthContext: The Case for Drug Repurposing
Cancer remains one of the leading causes of death globally. Conventional treatments such as chemotherapy, radiation therapy, and targeted agents are frequently limited by significant toxicity, high cost, development of resistance, and variable long-term efficacy.
In this context, drug repurposing has gained substantial attention as a strategy to rapidly identify effective and affordable therapeutic options — using medications with good patient tolerance and well-established safety profiles. This approach leverages decades of existing safety data to accelerate the development of new cancer therapies or adjunct treatments.
Important Caveats
The Authors' Own Assessment
"Given the observational design, reliance on self-reported outcomes, and potential for selection bias and uncontrolled confounding, these findings should be interpreted as hypothesis-generating. Urgent prospective, randomized, placebo-controlled clinical trials are warranted to validate these observations and further define optimal dosing strategies."
Peter Gillooly, CEO of The Wellness Company, framed the motivation behind the analysis: "Cancer patients come to our renowned doctors by the thousands for recommendations on how to handle their diagnosis. The American people, particularly when treating devastating illnesses like cancer, deserve answers supported by real-world data."
Featured Product
Research Authors
Media Inquiries
To arrange interviews with Dr. Peter McCullough, Dr. Kelly Victory, Dr. Drew Pinsky, Dr. Harvey Risch, or other authors, contact Chris Barron at cbarron@rightturnstrategiesdc.com.
This article is for informational purposes only and does not constitute medical advice. Ivermectin and mebendazole are used here in an off-label capacity. Always consult a qualified healthcare provider before making decisions about your treatment. The manuscript referenced is currently under peer review and has not yet been published in a peer-reviewed journal.
Because the research constitutes a public health surveillance activity,
the study did not constitute human research and does not require institutional review board review
or exemption according to the Common Rule (45 CFR §46). We followed the reporting guideline for
case series studies.






















